Remember the 3 large distinctions of anemia types and potential etiologies:
Microcytic (MCV < 80)
Normocytic (MCV 80-100)
Macrocytic (MCV >100)

Know how to interpret an iron panel in a medically ill patient:
1) Ignore the transferrin saturation
- Reason: does not discriminate between anemia of inflammation and iron deficiency
2) Assess ferritin
- <20: Iron deficiency
- 20-100: Iron deficiency likely if inflammation, chronic viral infection, etc.
- 100-800: Unlikely iron deficiency (unless hepatitis, chronic HD)
- >800: Extremely unlikely iron deficient
3)Assess the TIBC (preferred over transferrin level)
- >400: Iron deficient
- 300-400: Likely a component of iron deficiency
- 200-300: Unlikely iron deficient
- <200: Very unlikely iron deficient, usually inflammation, liver failure
Tuberous Sclerosis:
- Autosomal dominant genetic disorder due to a mutation in either TSC1 or TSC2 gene.
- Incidence of 1 in 5000-10000 live births
- De novo mutations account for ~ 80% of TSC cases
- TSC is highly variable in expression – thus the severity of disease can vary substantially among affected individuals within the same family
- TSC is characterized by the development of a variety of benign tumors in multiple organs: brain, heart, skin, kidney, lung, and liver
81-95% of TSC patients have on the characteristic skin lesions:
- Angiofibromas ~ typically on the malar region of the face
- Hypopigmented macules AKA ash-leaf spots ~ elliptic in shape
- Shagreen patches ~ usually over the lower trunk
- A distinctive brown fibrous plaque on the forehead ~ usually the first and most readily recognized feature of TSC
- Periungual/subungual fibromas develop during adolescence or adulthood; toenails > fingernails
CNS lesions characteristic of TSC include:
- Glioneuronal hamartomas (corticol tubers)
- Subependymal nodules
- Subependymals giant cell tumors (SGCTs): the characteristic brain tumor of TSC with a prevalence of ~ 5-20%, with 6-9% symptomatic
Neurological complications include:
- Seizures ~ 79-90% of patients, most often in the 1st year of life
- Cognitive deficits ~ 44-65%, associated with a history of infantile spasms or refractory seizures
- Behavioral problems ~ 40-90%; typically hyperactivity, inattention, and self-injury
Cardiovascular complications include:
- Rhabdomyoma: a benign tumor that often presents as multiple lesions
- No evidence for malignant transformation; no treatment is necessary for asymptomatic tumors
- Unlike other lesions of TSC, cardiac rhabdomyomas often disappear in later life spontaneously
Renal complications include:
- Angiomyolipomas ~ 55-75% (estimated incidence)
- Benign tumors composed of abnormal vessels, immature smooth-muscle cells, and fat cells
- Due to the abnormal vasculature and potential for aneurysms, spontaneously life-threatening bleeding is a potential complication
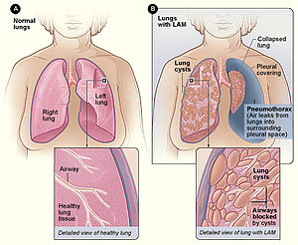
Pulmonary complication of TSC:
- Lympangiomyomatosis (LAM); Women (almost exclusively) – Widespread pulmonary proliferation of abnormal smooth-muscle cells and cystic changes within the lung parenchyma
- Two common intial manifestations of LAM: dyspnea and spontaneous pneumothroax
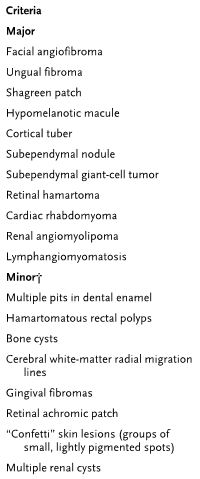
Diagnostic criteria for TSC:
Diagnosis is based on either genetic testing results and/or clinical findings
Genetic criteria:
- Identification of either a TSC1 or TSC2 pathogenic mutation (i.e. a mutation that clearly inactivates the function of TSC1/2 proteins)
Clinical criteria:
- 11 major and 6 minor features
- Definite diagnosis requires 2 major or 1 major/>2 minor features
Management of TSC:
- Requires a multidisciplinary approach of specialists including: neurologists, dermatologists, geneticists, and pulmonologists
- Referral to a TSC clinic is recommended given the complexity of the disease (next slide)
- Long-term follow-up including monitoring of lesion growth (angiomyolipomas and SGCTs); no conclusive guidelines exist
- Standard practice: brain/abdominal imaging every 3 years; more frequently with known lesions
- Genetic counseling for family planning (risk of affected child is 50%)
Therapeutic Options:
- Sirolimus (mTOR inhibitor) has been identified as a potential option given tumor cells from patients with TSC active mTOR (studies ongoing)
