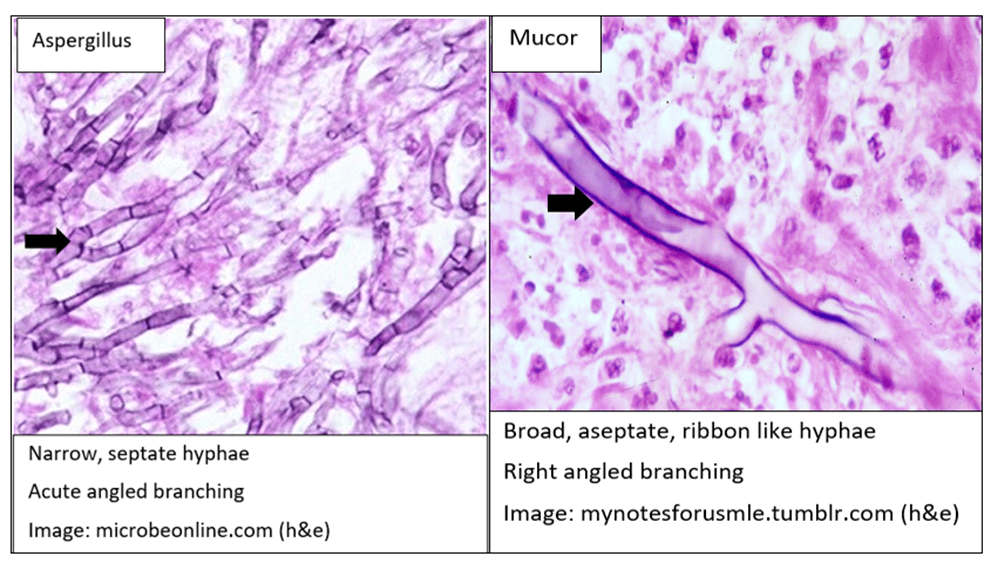
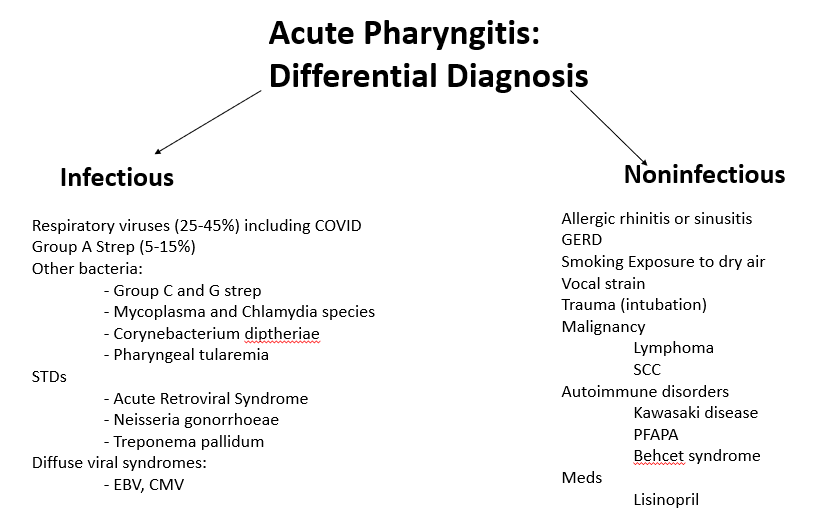
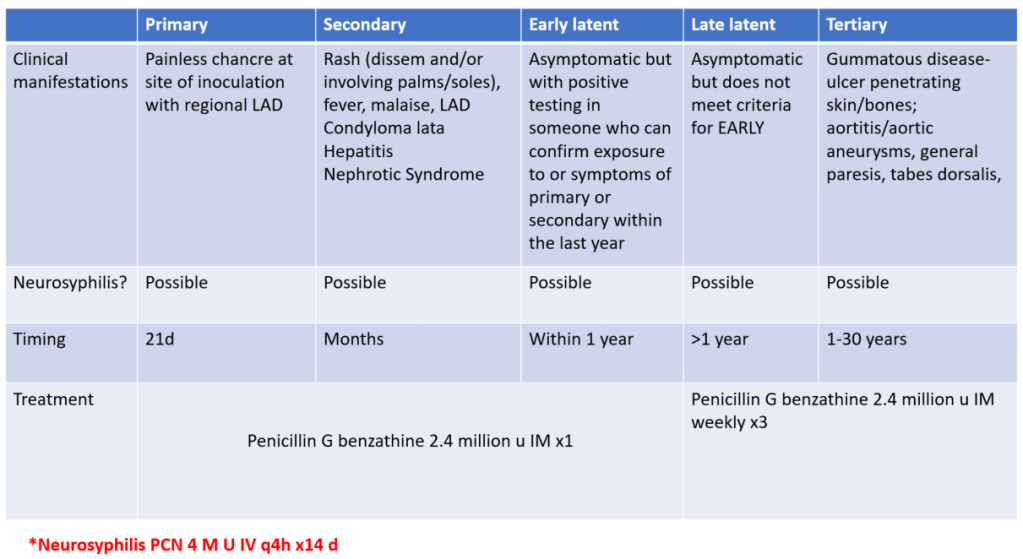
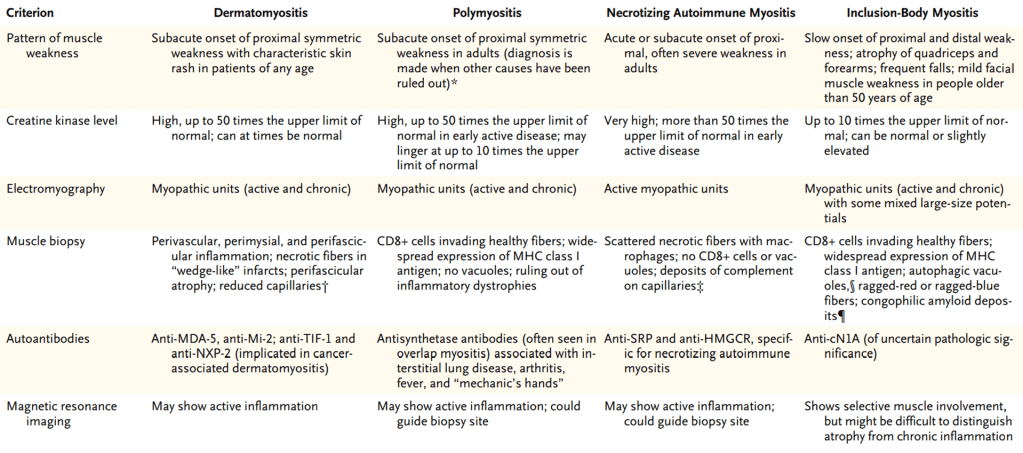
We discussed a case about an older man with chronic HCV (treated ~2 years ago) and 10-pack-year smoking history, who presented with acute fevers, rigors, SOB, cough, and was admitted for acute hypoxic respiratory failure due to Legionella pneumonia (Legionnaires’ disease).
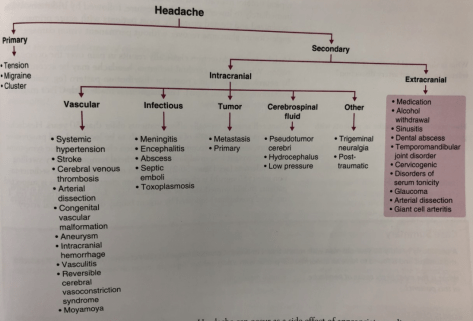
Severity of CAP
There are several severity scoring systems for CAP. Most common systems are the PSI / POST Score and CURB-65. The IDSA / ATS also has a criteria for defining severe CAP.
Highlights about the clinical utility of MRSA nares screening to rule out MRSA pneumonia
Legionella pneumonia (Legionnaires’ disease)
- Accounts for ~1-10% of CAP
- Patients can present as severe hypoxic respiratory failure. There are case series that report up to 44% of Legionnaires’ disease require ICU admission.
- In addition to outbreaks from aerosolized contaminated water systems, sporadic cases are quite common (causing the majority of cases) and the source of infection is often unknown.
- Older age, smokers / chronic lung disease, other chronic diseases (CVD, renal), and immunocompromised state are risk factors.
- Consider testing for Legionella in…
- All patients with moderate to severe CAP
- Patients with CAP who require hospitalization
- Any patient with CAP or nosocomial pneumonia who has a known or possible exposure to Legionella
- Immunocompromised patient (given high risk for Legionella and severe disease)
- Diagnosis of Legionella
- PCR of lower respiratory tract samples (ie BAL) is preferred because of high diagnostic accuracy and detects all Legionella spp (there are >60 Legionella spp!).
- Urine Ag is also acceptable. This only detects Legionella pneumophila serotype 1 (which is prevalent in the US).
- Treatment of Legionella
- Macrolides and fluoroquinolones
- Duration: ~5-10 days and do not stop therapy until patient is clinically stable and afebrile for at least 48 hrs
- Procalcitonin-guided deescalation is not well established in Legionnaires’ disease. Procal levels do rise in Legionnaires’ disease, but may not rise to the same degree as with pneumonia caused by other organisms such as strep pneumoniae.