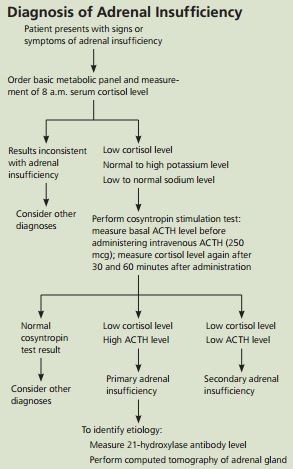
Surreptitious Insulin Use:
– Exogenous insulin intake is associated with increased insulin and low C-peptide levels
Whipple’s Triad:
1.Symptoms consistent with hypoglycemia
2.Documented low plasma glucose when symptoms are present
3.Relief of symptoms following resolution of hypoglycemia
– Consider a psychiatric evaluation in patient’s suspected of intentional exogenous insulin use

– Endogenous insulin is formed as two insulin chains (A&B) linked by C-peptide. Measurement of C-peptide can help distinguish from endogenous versus exogenous insulin
Insulinoma:
Diagnosis:
– Clinical + measurement of insulin (normal or elevated), proinsulin (normal or elevated), and C-peptide (normal or elevated)
– Localization of tumor with imaging; start with CT of the abdomen
|
Glucose |
Insulin |
Proinsulin |
C-peptide |
BHB |
| Exogenous Insulin |
↓ |
↑ |
↓ |
↓ |
↓ |
| Insulinoma, NIPHS, PGHS |
↓ |
↑/NL |
↑/NL |
↑/NL |
↓ |
| Sulfonylurea Ingestion |
↓ |
↑ |
↑ |
↑ |
↓ |
– Use a urine sulfonylurea toxicity screen to distinguish insulinoma from sulfonylurea ingestion
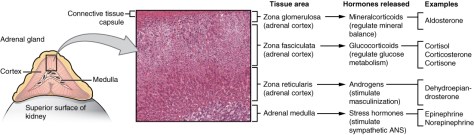
Pheochromocytoma:
– always think about pheo when the clinical case includes episodes of hypertension and headache
– Pheochromocytomas are tumors composed of chromaffin cells of the adrenal gland
– Pheochromocytomas almost always realease catecholamines
Classic Triad:
– Diaphoresis
– Headache
– Tachycardia
Genetic disorder associated with pheochromocytoma:
– MEN 2A and 2B
– Neurofibromatosis type 1
– Von Hippel-Lindau syndrome
Diagnosis: – Plasma / Urine catecholamines
– plasma high sensitivity (96-100%), but lower specificity (85-89%); 24 hour urine sensitivity/specificity (91-98%)
– Plasma will exclude a pheo when negative, but need to confirm if positive
– Following biochemical diagnosis – radiographic localization is needed
Preoperative management:
– Alpha-blocker – typically with phenoxybenzamine – is first-line therapy; followed by B-blockers (metoprolol, propranolol) to treat reflex tachycardia
MEN 1, 2A, and 2B disorders:
MEN 1: Pituitary adenoma, Parathyroid hyperplasia, Pancreatic tumors
MEN 2A: Medullary thyroid cancer, Parathyroid hyperplasia, Pheochromcytoma
MEN 2B: Medullary thyroid cancer, Marfanoid habitus/Mucosal neuroma, Pheochromocytoma

Cushing Syndrome:
– Bright purple abdominal striae always, always, always are associated with Cushing syndrome – striae larger than 1 cm in width are highly specific for hypercortisolism

Other classic stigmata:
– cervicodorsal fat pad aka “buffalo hump,” acne, hirsutism, moon facies, plethora, easy bruising, hypertension, insulin resistance, hypokalemic metabolic alkalosis, osteoporosis

Cause:
– Elevated cortisol
– Most common cause is exogenous glucocorticoid therapy for another cause
3 biochemical testing options for CS:
– 1 mg (low dose) dexamethasone suppression test – failure to suppress AM cortisol indicates true Cushing syndrome
– 24 hour urine free cortisol (UFC) – excludes CS when not elevated
– Late-night salivary cortisol
Never measure a random cortisol as part of the work up
After confirming CS biochemically, measure ACTH to establish etiology
– differentiate between ACTH-dependent/Cushing Disease (usually > 20 pg/mL) and ACTH-independent (usually < 5 pg/mL)
If ACTH-dependent CS: order pituitary MRI
If ACTH-independent CS: adrenal gland imaging (either CT or MRI)
ACTH-secreting pituitary adenoma is known as Cushing disease
Pearl: pituitary adenomas do not suppress with low-dose dex, but do suppress with high dose
Other possible ACTH-secreting tumors (but very rare):
– An ectopic ACTH-secreting tumor
– Usually these are primary lung cancers or a carcinoid tumor
– The clinical presentation of an ectopic lung tumor usually is less cushingoid (presenting with only HTN and metabolic abnormalities) due to the rapid growth of these tumors