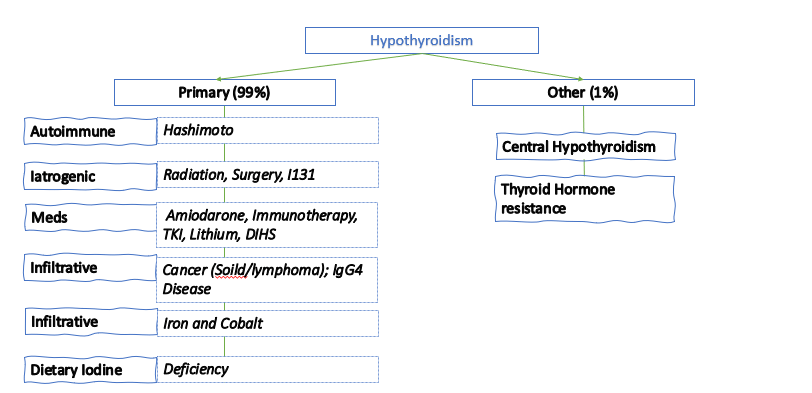
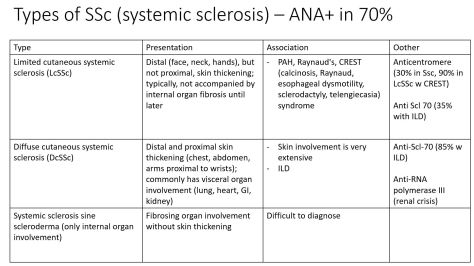
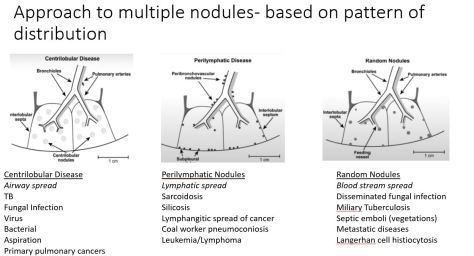
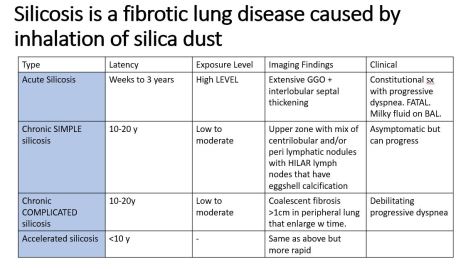
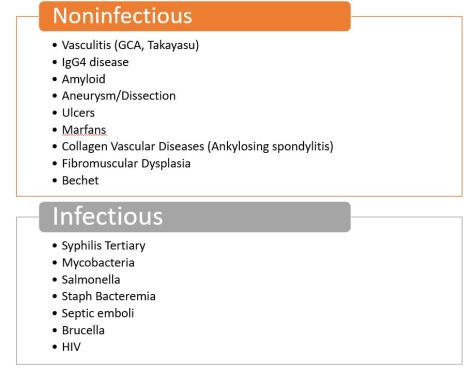
We discussed an interesting case of a 69yo female who presented with acute onset AMS and hypothermia diagnosed with myxedema coma with TSH 383 and free T4<0.1. She had been on treatment with Atezolizumab for high-grade urothelial cancer. Immune checkpoint inhibitors can cause multiple toxicities including hypophysis, siccca syndrome, thyroiditis, myocarditis, pneumonitis, ATN, AIN, MCD, GN, vasculitis, colitis and perforation.
While treating myxedema coma, always give glucocorticoids FIRST to avoid precipitating adrenal crisis in those with undiagnosed adrenal insufficiency. We also treat with thyroid hormone, supportive measures and treating underlying etiology.