Define:
Nephrotic Range Proteinuria: >3g/24 hours without other findings
Nephrotic Syndrome:
– Proteinuria > 3.5 g/24 hours (protein/creatinine > 3.5 mg/mg)
– Hypoalbuminemia
– Clinical evidence of edema
Secondary Causes of Nephrotic Syndrome:

Primary Nephrotic Syndrome:
Minimal Change Disease:
Epidemiology:
– Children < 10 years old; can be primary or secondary
– 10% of cases in adults
Clinical Features:
– Sudden onset of edema
– Thrombotic episodes more common in adults
– AKI may be seen in 20% at presentation
Diagnosis:
– Renal biopsy
MCD on light microscopy:
– Appears essentially normal (hence the name minimal change!); tubules may show lipid accumulation

MCD on electron microscopy:
– Characteristic fusion and effacement of podocyte foot processes

Focal Segmental Glomeruloscerlosis (FSGS):
Epidemiology:
– most common cause of primary nephrotic syndrome in the US
– can be primary, familial, or secondary
– African-Americans more common, but increasing incidence in all races
Clinical Manifestations:
– Asymptomatic proteinuria up to nephrotic syndrome ~ 2/3 at presentation!
– Hypertension usually seen in 30-50%
– Decreased GFR at presentation 20-30%
Diagnosis:
– Renal biopsy
FSGS on light microscopy: scarring or sclerosis involving some (focal) glomeruli, which are affected only in a portion of the glomerular capillary bundle (segmental)
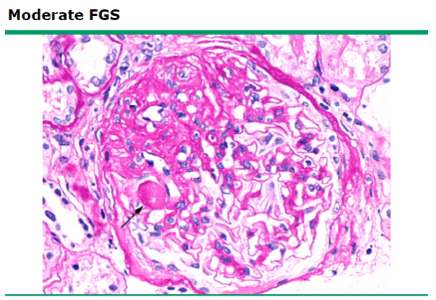
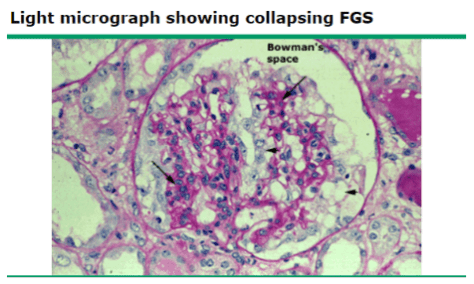
Collapsing FSGS – variant associated with HIV
Membranous Nephropathy (MN):
Epidemiology:
– most common in adults (>60 years old) and Caucasian
– can be primary (immune complex disease) or secondary (infection, autoimmune, cancer, drugs)
Potential complications:
– thrombotic disease – especially renal vein thrombosis
Diagnosis:
– renal biopsy
– PLA2R antibodies found in 75% of cases
Light microscopy for MN: diffuse thickening of the glomerular capillary wall

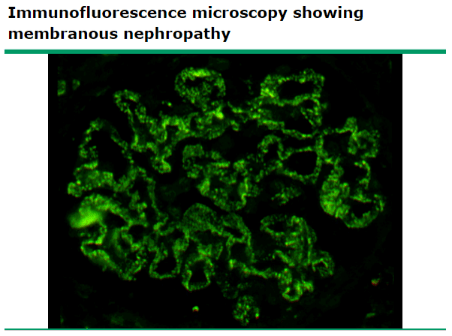
Immunofluorescence microscopy: diffuse, granular IgG deposition along capillary walls
Treatment:
– management is limited by a lack of clear evidence-based guidelines
General treatment:
– restrict dietary sodium to < 2 g/day
– restrict fluid intake to < 1.5 mL/day
Loop diuretics can be ineffective given that they are protein-bound and serum protein levels are reduced
Can add a thiazide diuretic and/or administer IV albumin bolus to improve diuresis
ACEi/ARBs – typicially used to reduce proteinuria although degree of benefit is unproven and evidence supporting routine use is conflicting
BP goal 130/80
Recent Cochrane review found no evidence to support the use of lipid-lowering agents in NS patients
Typically improves with resolution of disease
Corticosteroids are often used despite an absence of supporting evidence
Recent Cochrane review showed that combining alkylating agent (cyclophosphamide) with a corticosteroid has some short and long term benefits for MN
One exception – NS due to SLE – highly effective and supported by multiple studies