Narges presented a case of a late-middle age woman with history of recurrent pancreatitis, chronic alcohol use, who presents with worsening leg swelling, nausea/vomiting, and chest pain in setting of increased alcohol intake from bereavement. She increased her alcohol intake from “several drinks” a day to a fifth of hard liquor per day for the past few weeks. She was incidentally found to have a severely elevated Transferrin Sat, and genetics study revealed HFE mutation consistent with hemochromatosis.
For those of us illiterate in EtOH… A standard drink is defined as:

Hemochromatosis:
Epidemiology
- Hereditary: Up to 10% of Caucasians in US and Western Europe are thought to be heterozygous. Homozygous roughly 0.5%.
- Symptomatic median age around age 40 for males, later for females due to higher iron loss.
Pathophysiology
- Disorder of iron storage, that results in increased intestinal iron absorption and iron deposition.
- Iron deposition leads to organ damage.
- Hereditary form
- Most commonly secondary to mutant HFE (human hemochromatosis protein).
- HFE codes a protein involved in cellular iron sensing and intestinal iron absorption regulation.
- Autosomal recessive with variable penetrance.
- Heterozygotes are asymptomatic and are not at risk of iron-overload.
- Homozygous patients have variable disease penetrance, thought to be relatively low.
- ***Key point: Just because someone has the mutation (can be incidentally found on genetic sequencing) does not mean they will have the disease (iron build up in the body leading to end organ damage).
- Alcohol intake is a major risk factor for development of liver disease for patients with HFE mutation. Iron overload thought to potentiate effect of alcohol induced liver toxicity
- Liver damage occurs without inflammation, but hence HH can occur in setting without elevated AST/ALT.
- At least 43% of HH pts have other underlying causes i.e. fatty liver, alcohol, HBV/HCV, that leads to elevated transaminases and liver cirrhosis.
- Most common mutant is C282Y, if heterozygous, not at risk for developing progressive or symptomatic iron overload.
- Homozygous C282Y are at risk but again penetrance is variable.
- Homozygous H63D are generally not at risk but might have minor abnormalities in iron studies. If symptomatic, usually has other underlying process i.e. alcohol (such as this patient).
- Secondary iron overload (seen in frequent transfusions due to ineffective erythropoiesis)
Presentation
- Takes time to develop end-organ damage. Patients are usually asymptomatic until they have 20+ g of iron built up in the body (average iron content of an adult is 4-5g).
- Early: Non-specific, weakness, weight loss, skin hyperpigmentation (bronze skin), abd pain, loss of libido.
- Later: Hepatomegaly (95%), cirrhosis, HCC
- Other organs:
- DM (50%, pancreas) chronic pancreatitis (advanced hemochromatosis)
- Hypogonadism (ovaries, testes), impotence/infertility
- Hypothyroidism (thyroid)
- Adrenal insufficiency (adrenals)
- Arthralgia (joints, CPPD pseudogout), osteoporosis (pan hypopit leading to secondary hypogonadism)
- Heart (heart failure, enlargement)
- Pan-hypopit (pituitary)
Infection Risk
- Listeria, Yersinia, and Vibrio sp have increased in an iron rich environment. Mucor sp also favors an iron-rich environment
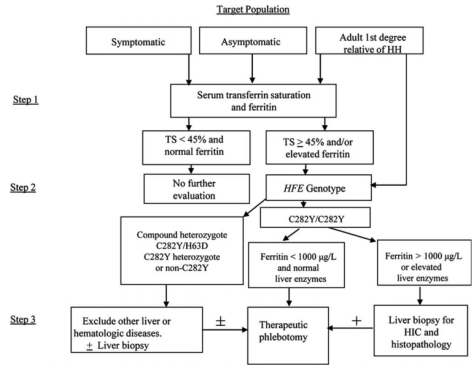
Diagnosis
- Labs: Inc Fe, % transferrin sat, and serum ferritin levels.
- Healthy pt: Fasting serum transferrin sat > 50% is abnormal and possibly suggestive of underlying hemochromatosis.
- Serum transferrin sat: PPV of 26-39%, 80% if two separate, positive tests. NPV of 100%
- Ferritin levels: not that useful, acute phase reactant, can fluctuate.
Management
- Remove excess iron!
- Intermittent phlebotomy, goal serum iron 50-100mcg/dL
- 1 unit of blood = 250mg Fe, hence to get 20g of iron town to a relatively normal level via phlebotomy, this might take 2-3 years.
- Chelating agents: Deferoxamine, less effective compared to weekly phlebotomy strategy but indicated in instances when you cannot do phlebotomy, i.e. anemia.
Prognosis
- IF caught early, organ damage is reversible with iron removal
- If cirrhosis has set in, this increases risk of hepatocellular carcinoma by 20-200 fold.
Screening
- All 1st degree family members of a patient with confirmed hemochromatosis should be screened with a genetics study +/- an iron panel.
- Per AASLD 2011 Guideline: