Thank you everyone for coming to our monthly M&M Conference!
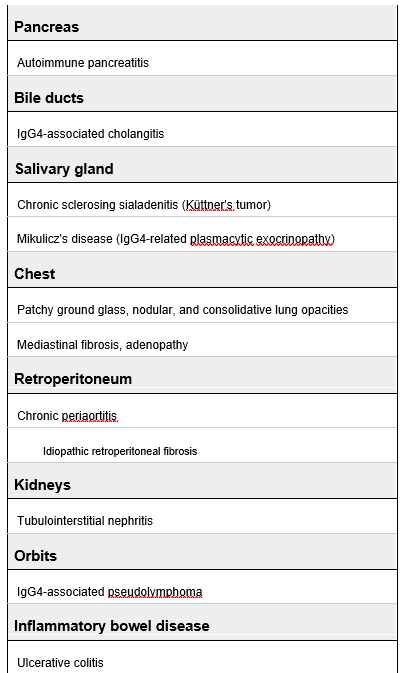
This was case of a young man with undisclosed chronic HBV and cocaine use who presented with first onset hematemesis and melena. On presentation, he was tachycardic, anemic, but normotensive. His heart rate improved with more fluid resuscitation and at the time, he did not require ICU level of care.
While on the floor, his tachycardia gradually worsened, and his Hgb also downtrended from 9.6 from 7.8 in the span of 12 hours, which was initially attributed to dilutional error given he was given 3 L of fluids. He acutely decompensated in the evening, and despite our best efforts, we could not keep up with the amount of hemorrhage likely from variceal bleeding.
Cocaine
Mechanism in a nutshell: Inhibits catecholamines reuptake, leading to to increased levels or norepi, dopamine, and serotonin. This subsequently leads to elevated BP, tachycardia, fever, and inc risk of coronary artery vasospasm, arrhythmia, and QTc prolongation.
Onset of action: Peak cardiovascular effect seen 5-15 minutes after use.
Half life: 45-90 minutes
Metabolism: Metabolized in seurm and liver into ecgonine methyl ester and benzoylecgonine, which is what Utox picks up! Utox will be positive for cotaine 48-72 hours after use, and it can remain positive up to 10 days (even longer for chronic users).
Cocaine & Anesthesia
- Compared to controls, pts who are cocaine positive on utox undergoing elective procedures requiring general anesthesia are no different in terms of:
- Arrhythmia risk
- Cardiovascular events (fluctuations in BP, HR)
- Anesthesia duration
- Medical dosage
- PACU recovery time
- This study in particular was insightful, but keep in mind that patients > 70, ESRD, or acutely intoxicated were excluded.
Hemorrhage Shock
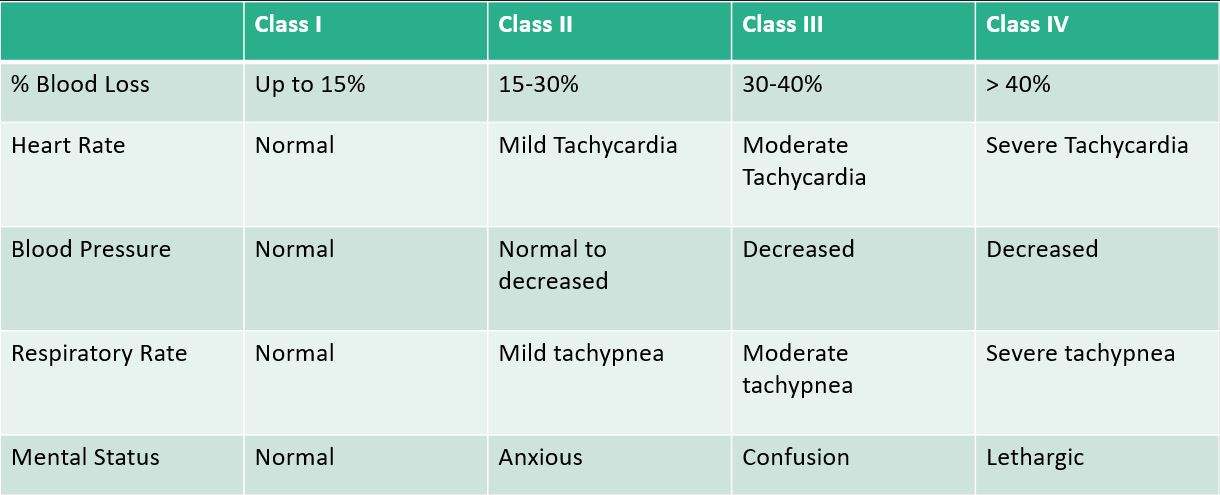
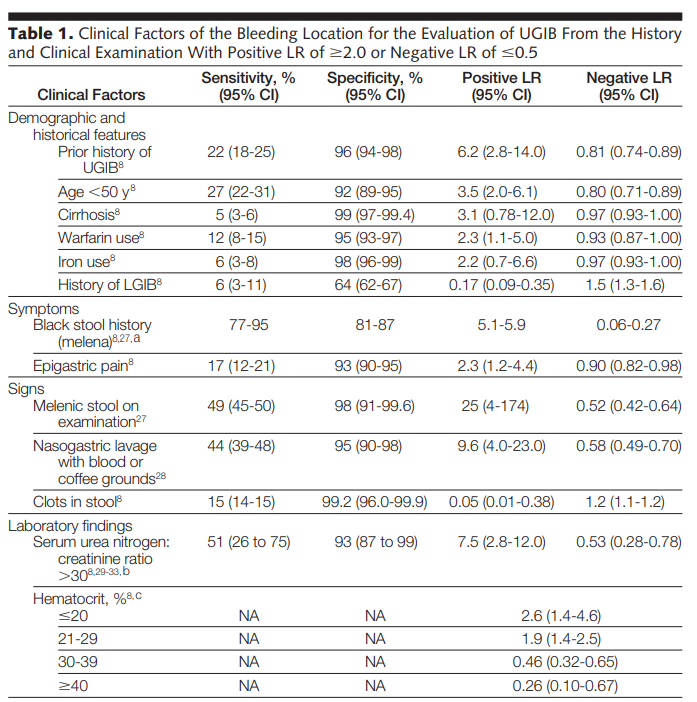
To put this into perspective, a patient (let’s say “Average” 70 kg adult) presenting with GIB can lose up to 1.5L of blood prior to seeing any chances in blood pressure. The first signs of clinical instability is HR, with mild tachycardia up to the lower 100s!
Also the Glawgow Blatchford Score is useful for risk stratification. If pt scores 0, there is a very low risk of complications. The higher the score is, the higher in terms of potential for complications and need for more urgent intervention.
When to Transfuse
See this study for more details. In general for UGIB, a restrictive transfusion strategy (transfuse when Hgb < 7 g/dL) has better outcomes (survival, rebleeding, mortality) vs a liberal approach (transfuse when Hgb < 9 g/dL).
In cirrhotic patients, is there such thing as over-resuscitation? YES.
In animal studies, severity of bleeding was directly related to portal venous pressure. Restrictive transfusion protocol also showed less portal pressure gradient vs the liberal arm, and less rebleeding and improved survival. Per the Baveno V Consensus, for variceal bleed, the target Hgb is between 7-8 g/dL.
What about when stuff hits the fan?
ABC comes first, don’t even worry about the numbers any more.
Remember when you order blood, it needs to be typed, screened, and cross-matched. It can take hours!
Know when to call the massive transfusion protocol (MTP). You can find the protocol on HHS-Connect on any desktop.
In a nutshell, activate MTP if you have a patient you anticipate requiring > 4 units PRBC within the first hour, or with high probability of requiring > 10 units within 12 hours. The blood bank will then start bringing in uncrossed blood, platelet, and FFP very quickly. Make sure to deactivate the protocol once your patient has been stabilized.
Also plan ahead for access! AT LEAST 2x large bore IV (18g or bigger) but know that in an acute situation, two PIVs can be easily overwhelmed. If you anticipate risk for decompensation, establish plan for access early on so you won’t get overwhelmed when the patient decompensates (i.e. IO kit or find an opportunity for a Cordis catheter).