Narges presented a very complicated patient, who presents with 2 days of abdominal pain and melena. He had a PCI 4 days prior with a drug-eluting stent deployed at the LAD, and he was discharged on aspirin and ticagrelor (PLATO trial, superior to clopidogrel). On presentation he was in hemorrhagic shock, with notable hematochezia on exam, encephalopathy, and a 4/6 systolic murmur with carotid radiation which turned out to be an undiagnosed severe aortic stenosis.
To briefly summarize his main problem list:
- Hemorrhagic shock secondary to UGIB
- CAD with recent NSTEMI s/p DES to LAD x1 on DAPT
- Severe aortic stenosis, newly diagnosed
Question is, what to do if you see a patient like this? Let’s break it down.
Antiplatelet and PCI
- Drug eluting stents (DES) are designed to have lower rates of late re-stenosis than bare metal stents (BMS), but they are at higher risk for in-stent thrombosis due to delayed endothelialization.
- Some terminology:
- Stent re-stenosis:
- Gradual narrowing of the stent segment, usually occurs 3-12 months after stent placement.
- Can present as recurrent angina vs full blown MI
- Stent thrombosis
- Abrupt thrombotic occlusion of a previously patent stent, usually catastrophic MI
- Duration of DAPT? It depends! Per the 2016 ACC/AHA guideline

In a nut shell:
Elective PCI in stable CAD:
- BMS: At least 1 month of DAPT
- DES: At least 6 months of DAPT
Exact duration is yet to be determined; there is some evidence of decreased stent restenosis (DAPT trial) with longer duration of DAPT but at the expense of all cause mortality and bleeding complications. There are also newer studies (i.e. ARCTIC Interruption) that found no benefits with longer duration of DAPT.
For PCI in setting of ACS, however, the guideline is different:
Anti-platelet duration in setting of ACS
- Medical Therapy: At least 12 months of DAPT
- PCI (BMS or DES, doens’t matter): At least 12 months of DAPT
- CABG: Also at least 12 months of DAPT
In patients with high bleeding risk or personal history of bleeding and/or long term NSAID use, preemptive prophylaxis with a PPI can be considered. If bleeding risk is significant, you can potentially decrease DAPT duration to at least 6 months, on a case-to-case basis.
Now keep in mind our patient has a DES deployed 4 days ago and he’s now bleeding…
GI Bleed
Risk Factors
- Alcohol
- NSAID
- Anticoagulation
- Cirrhosis
- Cancer
Presentation
- UGIB: BUN/Cr ratio > 30 usually (not sensitive but specific), melena (gut transit of blood)
- LGIB: bright red hematochezia, lower BUN/Cr ratio, but do not be fooled, can be seen in brisk UGIB (especially in this patient).
Management
- Airway protection
- Large bore IVs (18 G), Cordis, IO (they work wonders in a pinch!)
- Fluid resuscitation
- Anti-acid (i.e. Protonix)
- Octreotide if suspecting variceal bleed, if catastrophic variceal bleed, consider deployment of a Blakemore/Minnesota tube
- Antibiotics for primary SBP prophylaxis if cirrhotic
- Blood product administration
- GI consultation, urgency depending on clinical status, EGD
High risk features on Endoscopy
The patient underwent EGD and a duodenal ulcer that was actively bleeding was found. There are certain features of an ulcer that we can use to risk stratify and determine how likely intervention will be successful, and how likely the ulcer will rebleed. This is the Forrest Classification, first published in the Lancet in 1974. Our patient in this case is class 1B, and he ended by rebleeding twice requiring additional endoscopic intervention.

Source: Alzoubaidi D, Lovat LB, Haidry R. Management of non-variceal upper gastrointestinal bleeding: where are we in 2018? Frontline Gastroenterology Published Online First: 09 February 2018. doi: 10.1136/flgastro-2017-100901
Failure of endoscopic therapy
- Predictors: active bleeding at time of endoscopy, visible vessel, > 2cm, posterior duodenal ulcers, and gastric ulcers on the lesser curvature are associated with higher risk of treatment failure/rebleeding.
- First line: 2nd endoscopic intervention
- If recurrent bleeding persists, surgical options or IR embolization should be considered
The key question here is, when to resume DAPT? The decision will have to be individualized and most would recommend restarting ASAP as soon as patient is clinically stable… At the same time, pt has one other newly diagnosed problem that makes his management tricky…
Source: grepmed
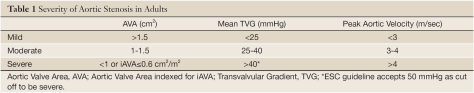
Severe Aortic Stenosis
Criteria:
- Area < 1cm2
- Mean trans valvular gradient > 40 mmHg
- Peak velocity > 4m/s
Referral to Cardiology because it is actually more complicated than this, there is also pseudo severe where AVR is not recommended, Low flow AS, low gradient AS, or both LF LG AS.
- Pseudo-severe: mild to moderate AS, low gradient, underlying myocardial dysfunction leading to poor valve opening. No benefit in AVR
- LF LG AS:
- AVA < 1cm2, but gradient < 35mmHg
- Must distinguish from pseudo severe AS. Do dobutamine stress echo
- If AVA remains the same, gradient inc, confirms true severe AS
- If AVA improves while mean gradient remains the same, this is pseudo-severe AS, manage medically.
Epidemiology
- < 70: Suspect bicuspid aortic valve
- > 70: Progressive valvular fibrosis/calcification
- Developing country: Rheumatic fever
Pathophysiology
- Over time leads to LV hypertrophy (concentric), muscle hypertrophy. Leads to heart failure over time.
Presentation
- Asx
- Sx: SAD syncope, angina, dyspnea
Management
- TAVR (transcatheter) vs SAVR (surgical), TAVR is non-inferior (PARTNER A, PARTNER B, SURTAVI trials) but has other complications to consider,
- Can think about the transcatheter approach in high surgical risk patients.
In setting of hypotension
- Patients with critical AS are highly preload dependent:
- Cardiac Output (CO) = Heart Rate (HR) x Stroke Volume (SV). In patients with severe AS, CO is fixed due to physiologic limitations of a small outlet.
- Increasing HR can help with CO but you run the risk of increasing myocardial O2 demands…
- Also in terms of volume, pts with severe AS usually has a degree of diastolic dysfunction (decreased compliance due to a hypertrophied ventricle). As you can imagine, if you push too much fluids into a non-compliant system, back up can occur leading to pulmonary edema thus respiratory failure…
- Choice Pressors: no absolute contraindication to any but phenylephrine has been suggested in some anesthesia text books as first line
- Rationale: Pure alpha, increases DBP (diastolic blood pressure), which in turns inc coronary perfusion pressure (CPP = DP – PCWP (LVEDP))
- Won’t induce tachycardia (if anything induces a mild reflexive bradycardia which decreases myocardial O2 demands).
- Epi has been associated with higher incidences of arrhythmias