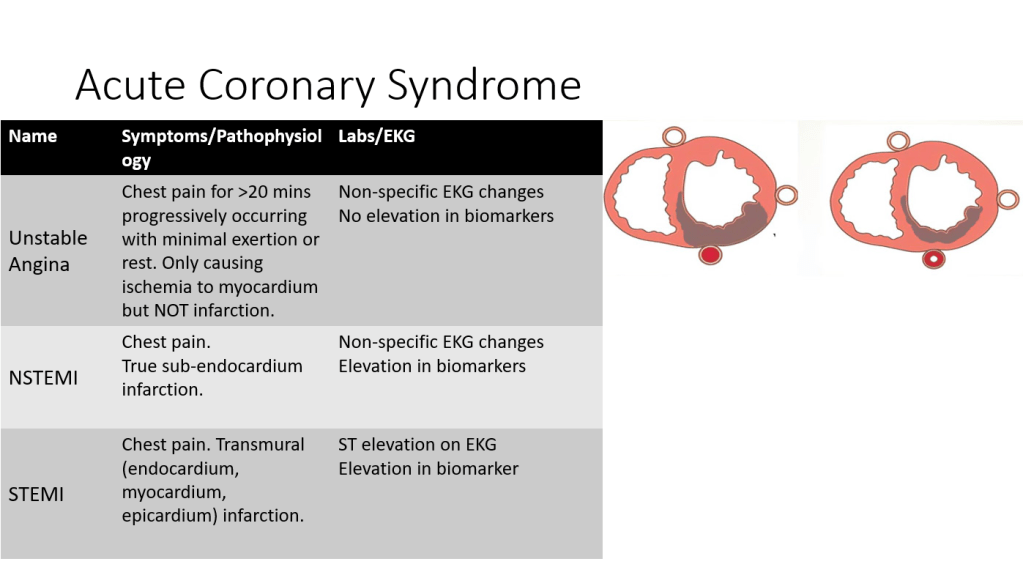
We discussed a case about an elderly woman with history of HTN, HLD, CVA, and rheumatic heart disease, who presented with palpitations and dyspnea on exertion, found to have severe mitral stenosis and atrial fibrillation. Unfortunately her hospital course was complicated by bilateral lower extremity arterial thromboemboli.
Rheumatic heart disease is the leading cause of mitral stenosis that affects females more than males (4:1).
Clinical manifestations
Patients can be asymptomatic for years until they develop severe stenosis.
- Dyspnea (due to pulmonary congestion)
- Fatigue (due to low cardiac output)
- LE edema (due to right heart failure)
- Orthopnea
- Pulmonary hypertension
- Systemic embolization
MS and atrial fibrillation
About 50% of patients with MS will have afib.
Without anticoagulation, these patients have a risk of thromboembolism of 20-25%.
Valvular afib = afib in the setting of…
- Moderate to severe MS OR
- Presence of a mechanical heart valve
Severity grading
- A: at risk of MS
- B: progressive MS
- C: asymptomatic severe MS
- D: symptomatic severe MS
Severe MS measurements:
- Mitral valve area ≤1.5 cm2
- Mean mitral valve gradient > 10 mm Hg
- Pulmonary artery systolic pressure > 30 mm Hg
Treatment
If the patient is a candidate, percutaneous mitral balloon valvuloplasty (PMBV) is the treatment of choice.
References
2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease