Gray Medicine had an interesting case of a 80yo F with history of treated TB (60 years ago), thoracic artery aneurysm s/p recent TEVAR, presenting with 3-4 months history of throbbing chest and back pain. She was admitted one month prior to the same complaint, CXR and CT Cx did not reveal significant pathology other than mild distal TEVAR graft dilatation. She presents 1 month later with worsening chronic chest pain, anorexia, weight loss.
This is the chest X-ray during this current hospitalization…
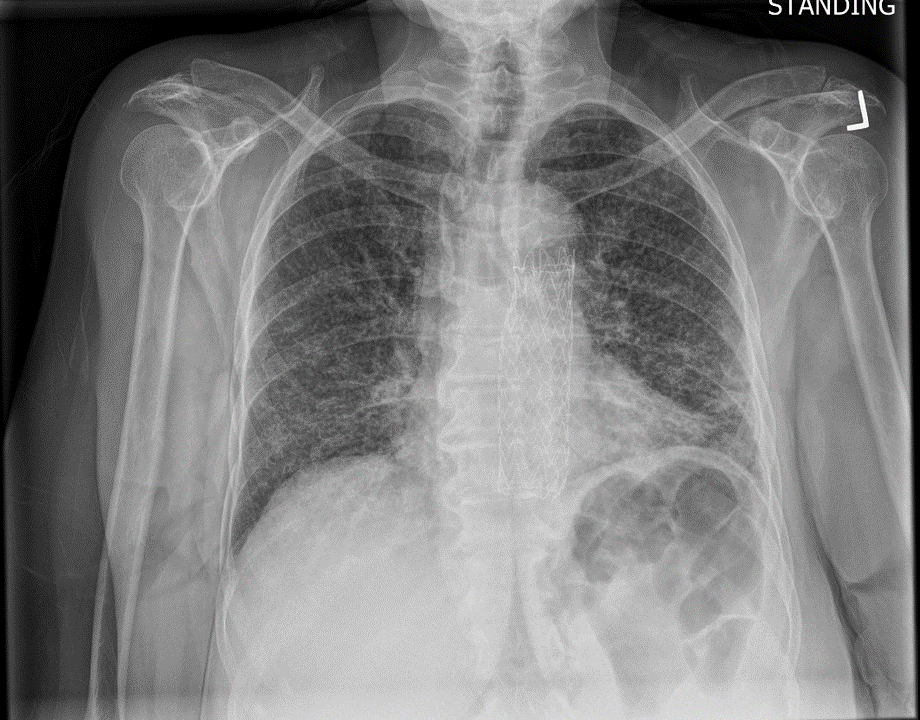
Burn this image into your head! This is a classic miliary pattern on a chest radiograph! The term miliary stems from millet seed, a term used to describe a group of small-seeded species of cereal crops or grains

Subsequent chest CT revealed innumerable bilateral pulmonary nodules, which were not present a month prior.
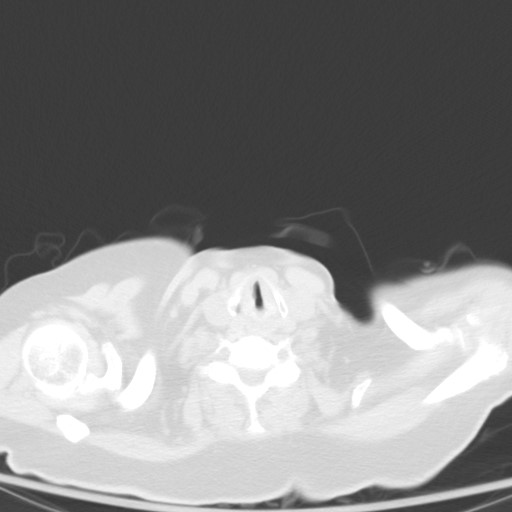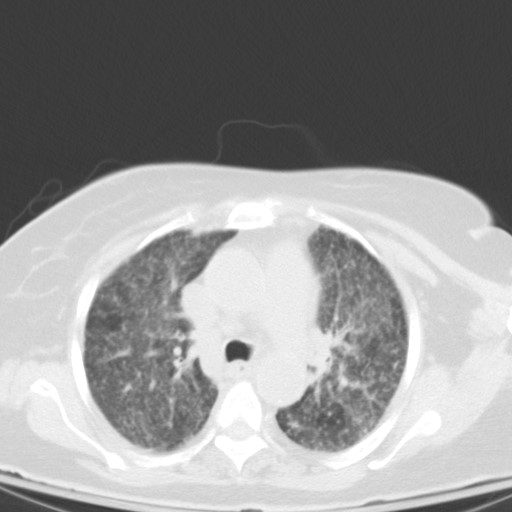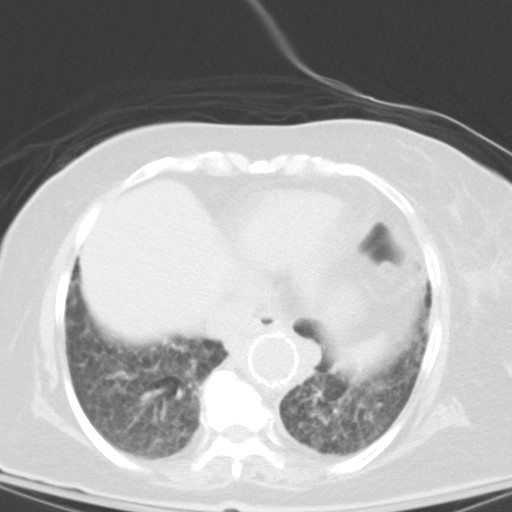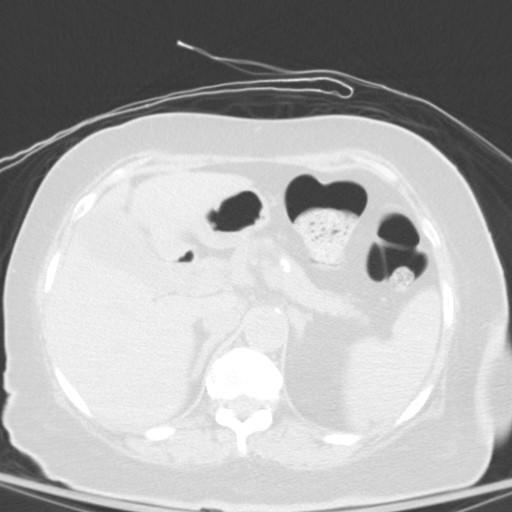
Let’s go through possible causes for a miliary pattern on a chest radiograph. In general it can be divided into three categories. The DDx can be quite wide!

In our case, given that our patient is an elderly woman with a remote history of treated TB, this presentation is highly concerning for miliary TB leading to reactivation. In general, miliary and disseminated TB are often used interchangeably. Disseminated TB refers to TB that affects at least two organ systems.
The most commonly affects organs are:
- Lungs
- Liver/GI
- Spleen
- Adrenals
- CNS
Our patient was placed on airborne, and ultimately her sputum was MTB PCR positive! She has TB!
Presentation of Miliary TB
- Very common: B-symptoms, FFT, typically subacute to chronic. 80-95% will have a fever.
- Miliary, unlike typical TB, can present with acute sepsis or respiratory failure.
- Pain/organ dysfunction based on location of the spread. Basically can affect anywhere. Hepatic TB, 79% of cases are due to miliary TB. Other commonly affected organs are spleen, adrenals, BM, lymphatics, and CNS.
- Other manifestations: DIC, hyponatremia, pan-cytopenia, 50% cases will have normocytic anemia.
Risk Factors
- Immunocompromised status
- HIV
- Extremes of age (infants, elderly)
- Immunosuppressives
- Post transplant
- Other medical co-morbidities (CKD, cirrhosis, EtOH, etc)
Diagnosis
- Chest radiograph: Classic faint reticulonodular infiltrate uniformly throughout lungs.
- CT is more sensitive for miliary TB and usually is recommended. Typical finding might reveal numerous 2-3mm nodules but this is not specific.
- Tissue, fluid, or lymph node biopsy
- Gastric aspirate
- Ultimately combination of clinical diagnosis with support labs/imaging.
- Gold standard: AFB and culture + MTB PCR
- All patient should have mycobacterial blood cultures
- Urine mycobacterial cell wall glycolipid lipoarabinomannan (urine LAM) is a highly specific test with high sensitivity in HIV patients for disseminated TB.
Management
- Intensive Phase: HREZ (aka RIPE) x 2 months
- R: Rifampin
- I: Isoniazid
- P: Pyrazinamide
- E: Ethambutol
- Continuation Phase
- After, 2 months of HREZ (RIPE), the continuation phase consists of 4 months of isoniazid and Rifampin.
- Choice of medication and duration will change depending on resistance of the organism and location affected
- Corticosteroids: Indicated if meninges or pericardium is involved.
- Make sure to fill out a GOTCH form (not the GOAT form, as someone answered on Kahoot this morning) for Santa Clara County if you have a patient with active TB since a safe dispo will involve multiple disciplines and careful planning!





