Thanks to doctor-in-training Emma who presented a case of an elderly woman, on dialysis, who came in with SEVERE lactic acidosis due to a variety of factors, but largely contributed by being prescribed metformin 2 weeks prior to presentation.
We will use this case to illustrate several concepts:
On initial presentation, pt was in a state of shock. If you remember from medical school, there are mainly 4 types:
- Cardiogenic
- Myopathic (DCM, acute MI)
- Mechanical (Acute MR, VSD, severe AS)
- Arrhythmia
- Hypovolemic
- Hemorrhage, volume depletion
- Distributive
- Sepsis
- Anaphylaxis
- Neurogenic (spinal cord injury)
- Endocrine (myxedema, Addison’s)
- Obstructive
- Pericardial tamponade
- PE
- Pneumothorax
Shock can also be approached from a cardiac output standpoint to help differentiate the potential causes:

If someone is hypovolemic, but he/she is found to be hypotensive but bradycardic (instead of tachycardic), remember from physiology:
MAP = CO x SVR, where CO is cardiac output, SVR is systemic vascular resistance
CO = SV x HR, SV is stroke volume, and HR is heart rate.
Hence in a state of hypovolemia, the body will attempt to maximize CO by increasing HR to maintain adequate MAP. If you see the opposite though, there could be something else going on…
- Cardiac: MI, conduction abnormalities, infectious (Lyme, Charga’s)
- Toxic/Meds: CCB, Beta blocker, Dig, Alpha 2, organophosphate
- Lytes: Hyperkalemia -> heart block -> sinus arrest
- Endocrine: Hypothyroidism (myxema coma), adrenal insufficiency
- Environmental: Hypothermia
- Neurogenic shock
Acid-Base: Acid-base disorders can appear daunting, but the following algorithm can make things simpler if followed consistently. We will work through this patient’s acid/base disorder together. Her HCO3 is 6, AG of 52, with an ABG of 6.83/13/170.
- Look at the pH: < 7.4 automatically means some sort of acidosis is going on.
- Next look at the PCO2, if it is in the same direction as the pH (low PCO2 in this case, and low pH), then it is suggestive of a primary metabolic rather than a respiratory issue causing the acidosis. If pH is low and PCO2 is high, then there is high suspicions for a respiratory process driving the acidosis.
- Look at the Anion Gap, which in this case in 52. Go through your differential for AGMA.
- Calculate the Delta Gap. This basically is a way to assess whether a mix acid/base disorder exists after taking into account the AG. To help illustrate:
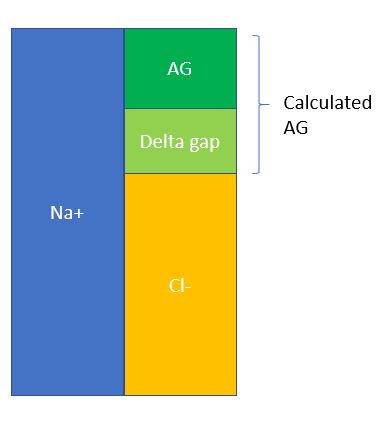
- Delta gap = calculated AG – normal AG = 52 – 12 = 40. Now there are several ways to do this, but one method that’s quick and simple is to simply add this delta gap to the measured bicarb and see what other metabolic acid/base disorders can be hiding beneath the anion gap. In this case, 40 + 6 = 46, which is suggestive of an underlying metabolic alkalosis.
- Lastly, calculate compensation. For AGMA, use Winter’s Formula = 1.5 (HCO3) + 8 +/-2. The calculated PCO2 should be 15-19. Since our measured PCO2 is 13, which is lower than expected, then this is suggestive of an underlying respiratory alkalosis as well, probably due to sepsis.
- Our final answer for the acid/base disorder for this patient is: AGMA with metabolic alkalosis and concurrent respiratory alkalosis!
Lactic Acidosis: Traditionally we view this as a sign of sepsis, or a marker of tissue hypoperfusion. However this is not always the case!
- Type A: Inadequate oxygen delivery
- Rule of thumb: Tissue hypoperfusion, tissue ischemia
- Can also be seen with increased anaerobic muscular activity, like generalized convulsions in a seizure
- Can also be seen in hypoxemia, severe anemia
- Cryptic shock (elevated lactate, okay BP), similar to overt septic shock in terms of mortality in setting of severe sepsis.
- Type B: altered physiology, others
- B1: Underlying disease, malignancy, hepatic & renal failure
- B2: Drugs, toxins
- B3: Inborn errors of metabolism, enzyme deficiency
- Management: Tx the underlying cause, bicarb controversial, studies have shown no benefits? But most experts would agree with usage in critically ill patients with profound acidemia.
MALA: Lastly, this case illustrates the dangers of metformin, in the right setting, can lead to severe acid/base disturbance (MALA).
- Do NOT use metformin if GFR < 30, be careful in CKD patients.
- If suspecting metformin toxicity and pt is hyperglycemic, this is a sign of severe toxicity.
- Always do a thorough med reconciliation on every single patient!




 Image adapted from Derm 101
Image adapted from Derm 101